Our research interests bridge the traditional disciplines of organic and inorganic chemistry. Catalysis and metal-mediated organic synthesis are the principal themes of our research program. We are developing the synthesis and reactivity of inexpensive, earth abundant, and environmentally benign base metal complexes that are of interest as catalyst precursors for applications in organic synthesis, lignin valorization, and polymer chemistry, as well as for modeling of catalytic reactions. These innovations advance transition of the chemical and materials industries towards more sustainable, energy efficient feedstocks and processes. Current projects include:
Development of Base Metal Catalysts for Carbon-Oxygen Bond Activation
- Catalytic Dehydration of 1-Phenylethanol to Styrene:
Global production of styrene is estimated at ~36.5 million tons in 2025, with 1-phenylethanol (PHE) dehydration to styrene accounting for ~15% of global production. However, PHE dehydration to styrene remains a significant challenge since it is currently accomplished via energy-intensive gas-phase processes carried out at ≥ 250 °C using, for example, titania or alumina LA catalysts. Moreover, high reactivity of the intermediate carbocation (equation 1) makes realization of high styrene selectivity a challenge.
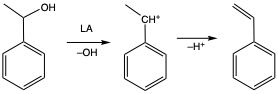
Advances in PHE dehydration catalysis are therefore critically needed to reduce energy requirements as well as improve selectivity for styrene. In this regard, we are developing zinc- and aluminum-based Lewis acid catalysts for selective dehydration of PHE to styrene that operate under more sustainable conditions.
- Conversion of Cellulose into 5-Hydroxymethylfuran
Furan derivatives, such as furfural and 5-hydroxymethylfuran (5-HMF), can be obtained from renewable carbohydrate biomass, and are highly attractive as sustainable chemical platforms for the production of fuels and chemical intermediates. In particular, 5-HMF could play a key role in the establishment of a biomass-based chemical platform since it can readily be converted into a variety of useful acids, aldehydes, alcohols, and amines, as well as the promising liquid fuel 2,5-dimethylfuran (2,5-DMF). We are exploring the development of environmentally benign catalytic processes for the conversion of lignocellulosic biomass into 5-HMF.
Preparation and Applications of Bronsted- and Lewis acidic Deep Eutectic Solvents (DESs) in Catalysis
Lewis acid (LA) catalyzed carbon-oxygen (C–O) bond activation is central in organic chemistry and materials science. However, because many conventional LAs are sensitive to water, there remains a critical need for the development novel water-tolerant LA catalyst systems to extend further utility of Lewis acid catalysis. In this context, ternary deep eutectic solvents (DESs) consisting of choline chloride (ChCl), hydrogen bond donor (HBD), and base metal salts are highly promising Lewis acid catalyst systems for lignin valorization to produce aromatic monomers and dimers, and renewed lignin for bioplastics and biofuels production. Their high potential is attributed to several factors, including (1)strong Lewis acidity and high tolerance to water of many base metal ions, and (2) deep eutectic solvents, formed from combination of ChCl (hydrogen bond acceptor, HBA) with appropriate HBD, are excellent at competing with the OH groups in lignin to disrupt its hydrogen bonding network, thereby solubilizing it and enhancing reactivity of its weak ether (C–O) and carbon-carbon (C–C) bonds. Thus, we are synthesizing base metal salt/ChCl/HBD ternary DESs and developing their efficacy as catalysts for lignin valorization to produce valuable products such as phenolic monomers and dimers, and renewed lignin that can be utilized in production of biofuels and bioplastics. Furthermore, we are investigating and elucidating significant knowledge gaps that remain in understanding of the structure-property-function relationship of ternary ChCl/HBD/metal salt DES systems.
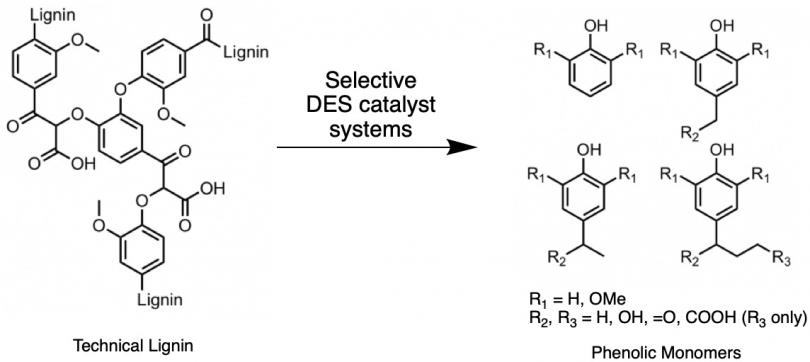
Synthesis and Reactivity of Zinc-based Frustrated Lewis Pairs (FLPs)
Catalytic hydrogenation to reduce or saturate organic compounds is ubiquitous in organic synthesis and industry. However, base metal hydrogenation catalysts are largely underdeveloped, as they are often much slower and/or require higher temperatures than late transition metal catalysts. Frustrated Lewis pairs (FLPs) are intra- or intermolecular combinations of Lewis acids (LA) and Lewis bases (LB) wherein dative bonding between LA and LB is sterically impeded (i.e., frustrated), or where separation of the LB donor and LA acceptor is transiently masked by a dative interaction.
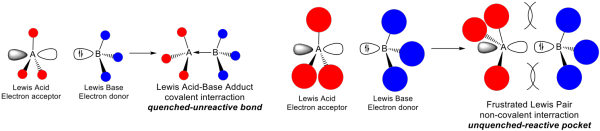
Several FLPs, especially P/B and N/B FLPs, exhibit very good to excellent ability to activate H2 and other small molecules, as well as serve as active catalysts for the hydrogenation of an increasing variety of unsaturated compounds, such as imines, enamines, aldehydes, ketones, olefins, and alkynes. However, while Lewis acidic boranes are quite effective at H2 cleavage, hydride delivery to a substrate for hydrogenation is slowed by the relatively strong B-H bond strength. We are designing and synthesizing novel Zn2+/LB FLPs and developing their efficacy in catalytic hydrogenation of unsaturated compounds, as well as for E-H (E = H, B, C, etc.) bond activation reactions.

